January 30, 2018 – Takara Bio’s Board of Directors resolved today to establish a new facility for the research and manufacturing of regenerative medical products in coordination with the expansion of existing research and manufacturing facilities at the company’s head office site in Kusatsu, Shiga.
Total project cost is expected to be around 7.3 billion yen, which is payable by cash on hand.
Takara Bio constructed a Center for Gene and Cell Processing in 2014 and the main building (new research building) at its Kusatsu facility in 2015, where it manufactures investigational drugs for its gene therapy clinical development projects and advances its regenerative medical product contract development and manufacturing organization (CDMO) business for regenerative medical products.
Gene therapy clinical development projects have been progressing smoothly and have now reached a stage where manufacturing can be scaled up in anticipation of market launch. Also, the CDMO business has seen an increase in contract service orders resulting from the effects of an increase in regenerative medical product development projects by pharmaceutical companies, bio-venture enterprises, and the like, and the expansion of the genome analysis diagnostic field, and the utilization rate of each facility has risen rapidly.
In consideration of this situation, Takara Bio has decided to establish a new facility in order to further advance its gene therapy clinical development projects and expand and develop CDMO business.
The new facility will be structured to facilitate contract service and R&D operations in three areas – the manufacturing of GMP/GCTP compliant regenerative medical products, quality testing, and operations such as genome editing and iPS cell manufacturing. With just the area with manufacturing equipment installed, the expansion enabled by the new facility will provide by approximately 2.5 times the contracted service sales recorded in fiscal 2016.
The new facility will have a total floor space of 14,100m2, and approximately 4,600m2 of this will not have manufacturing equipment installed, as it is designed to be used flexibly to respond to a diverse range of needs that may occur in the future. Furthermore, some functions from existing facilities will be transferred to and expanded in the new facility, and floor space in the existing facility for cell processing, cell bank storage, and gene analysis by a high-speed sequencer will be expanded. This plan will improve these functions and increase capacity.
Takara Bio will use this establishment and expansion of facilities as an opportunity to further develop and strengthen our operations according to the management objectives of “acquiring approval for the first gene therapy product in Japan” and “remaining number one for CDMO business involving regenerative medical products” as set forth in its Medium-Term Management Plan FY2020 (formulated May 9, 2017).
|
Establishment of a New Research and Manufacturing Facility and Expansion of Existing Facilities at the Head Office Site |
|---|
|
Address |
Nojihigashi 7-4-38, Kusatsu, Shiga, Japan |
|---|
|
Objectives |
|
|---|
|
Content |
(1) Construct new facility
(2) Existing facilities
|
|---|
|
Completion |
Scheduled for September 2019 |
|---|
|
Project expense |
Approximately ¥7.3 billion (total amount to be covered by Takara Bio) |
|---|
|
Primary construction companies |
|
|---|
Note: The above information applies to the plan at its current stage and may change going forward due to various factors.

Regenerative medical products
A separate category from pharmaceuticals and medical equipment established under the Pharmaceutical and Medical Device Act (effective November 2014). In addition to processed and prepared human cells, tissues, and other products traditionally used for transplants as part of regenerative therapy, it also includes gene therapy products and the like.
GMP/GCTP
GMP is an abbreviation for Good Manufacturing Practice and refers to the Ministerial Ordinance on Standards for Manufacturing Control and Quality Control for Drugs and Quasi-drugs that must be observed in the manufacture of pharmaceuticals. GMP requires controls for the manufacturing of pharmaceuticals and compliance with regulatory authorities in each country to safely provide pharmaceutical products of assured quality.
GCTP is an abbreviation for Good Gene, Cellular, and Tissue-based Products Manufacturing Practices and refers to the Ministerial Ordinance on Standards for Manufacturing Control and Quality Control for Regenerative Medical Products.
CDMO business
CDMO is an abbreviation for Contract Development and Manufacturing Organization. It refers to services involving the development of and production support for regenerative medical products.
Vector
Genetically disabled viruses used to transduce curative genes into cells during treatments such as gene therapy.
Center for Gene and Cell Processing
In 2014, Takara Bio completed construction on Japan’s largest regenerative medicine-related facility at the company’s head office site in Kusatsu, Shiga, with the purpose of manufacturing investigational drugs for its gene therapy clinical development projects and advancing its CDMO business. The three-story building has a total floor area of approximately 6,500m2, is GMP/GCTP compliant, and possesses approval for the manufacture of specific processed cells as stipulated by the Act to Ensure the Safety of Regenerative Medicine, etc. (effective November 2014) and ISO9001 certification. The facility won the Facility Integration category at the 2016 Facility of the Year Awards run by the International Society for Pharmaceutical Engineering, Inc. (ISPE), and is widely recognized as a facility that can realize the safe, efficient manufacturing of the various regenerative medical products required by regenerative medicine CDMO.
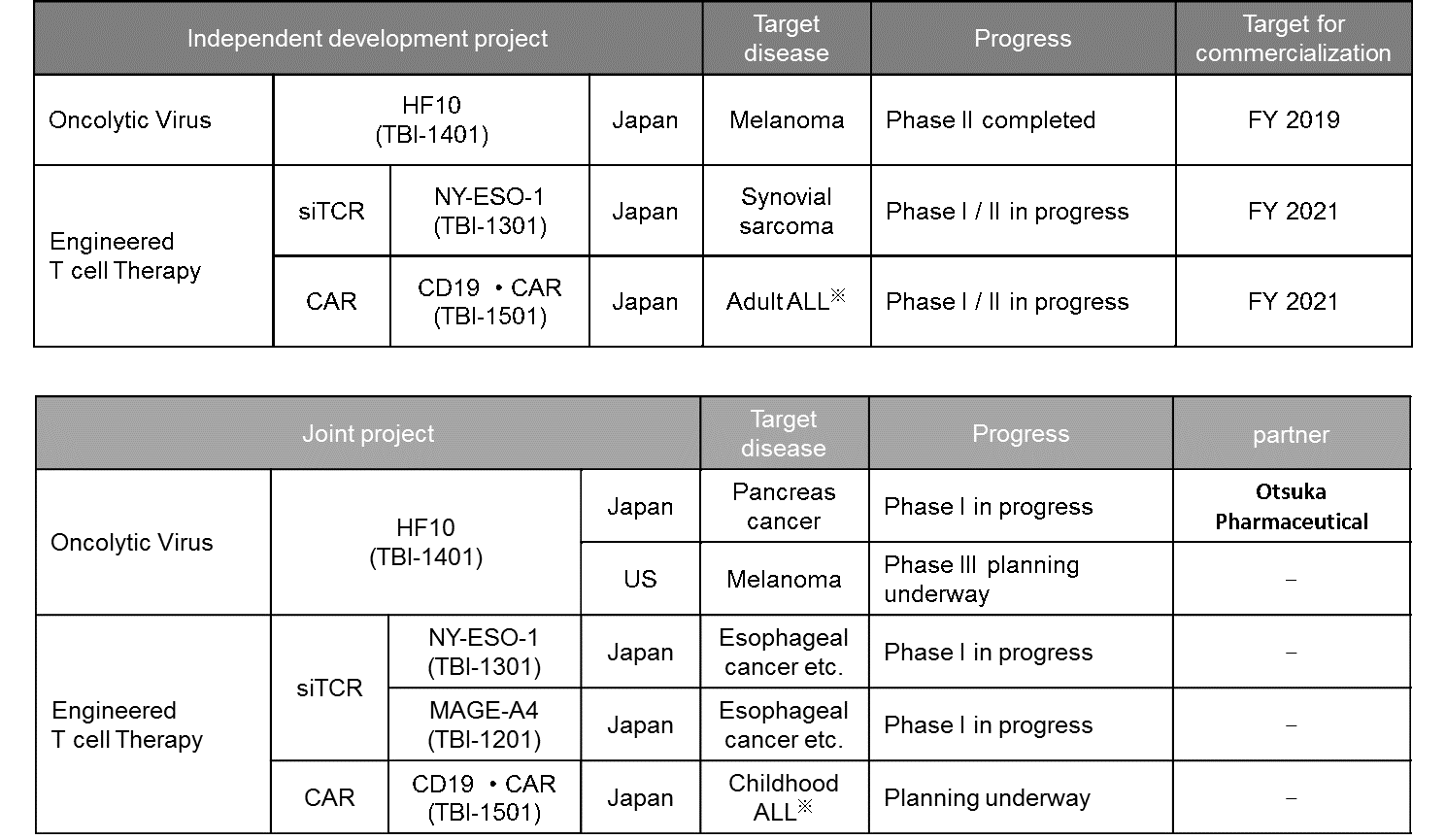
Overview of gene therapy projects being advanced by Takara Bio
Takara Bio is engaged in clinical trials involving oncolytic virus HF10 as a gene therapy for treating cancer, and genetically modified T-lymphocyte therapy CD19 CART, NY-ESO-1 siTCRTM). Please refer to the table below regarding the progress of our main projects.